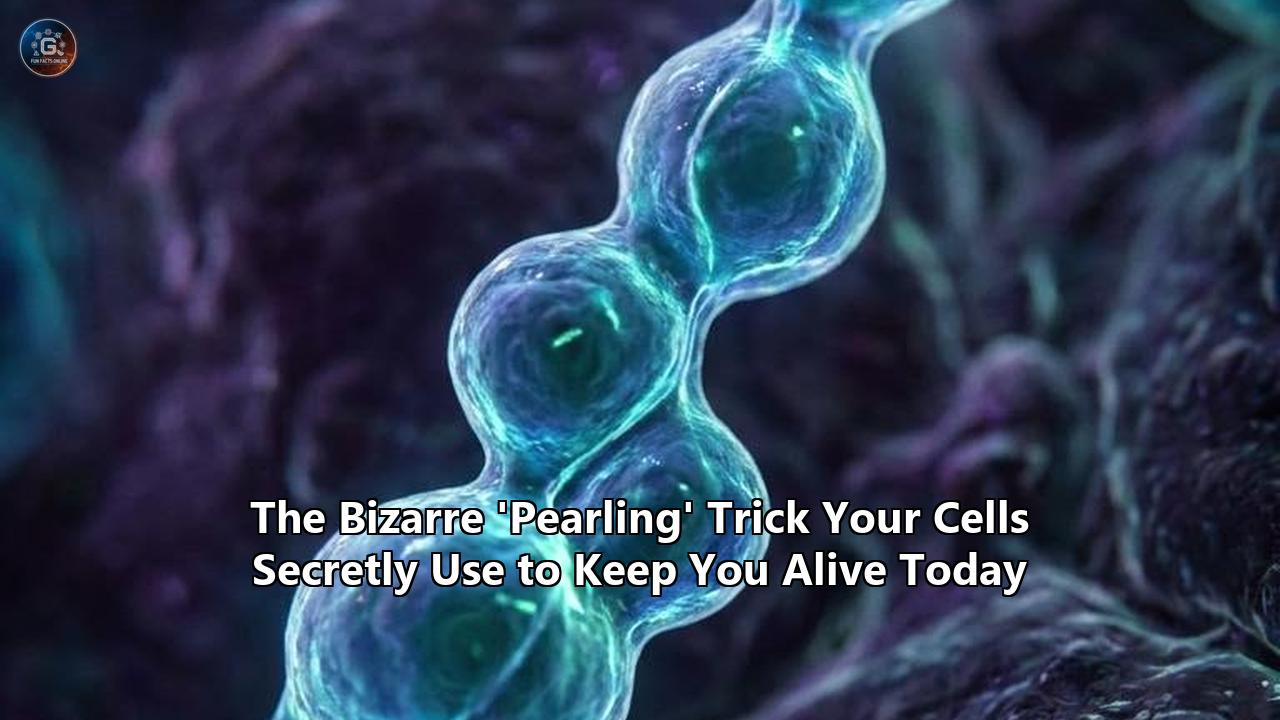
On April 2, 2026, researchers at the Ecole Polytechnique Fédérale de Lausanne (EPFL) published a finding in the journal Science that immediately forced a rewrite of standard cell biology textbooks. The team, led by biophysicist Suliana Manley and postdoctoral fellow Juan Landoni, demonstrated that mitochondria—the energy-producing organelles inside our cells—do not merely exist as smooth, static tubes. Instead, they undergo violent, transient physical contortions, shifting into a string of distinct, bead-like spheres multiple times a minute.
The EPFL researchers identified this phenomenon as "mitochondrial pearling." Far from being a random quirk of cellular anatomy, this bizarre morphological shift is a highly regulated, mechanical survival mechanism designed to evenly distribute mitochondrial DNA (mtDNA) across the organelle. When mitochondria form these pearl-like constrictions, they physically break apart clumping genetic material and force it into neatly separated compartments. Once the DNA is spaced out, the organelle relaxes back into its normal cylindrical shape.
This discovery anchors a broader, rapidly accelerating shift in how scientists understand cellular architecture. Over the past 24 months, high-speed imaging technologies have revealed that the cellular pearling process is not restricted to mitochondria. It occurs in the axons of our brain cells, in the formation of blood platelets, and even in the primitive structures of artificial, lab-grown cells. What biologists dismissed for over a century as a symptom of cellular death or toxic stress is actually a ubiquitous physical trick utilized by biological membranes to maintain structural integrity, manage chemical payloads, and keep organisms alive.
The Problem of Mitochondrial DNA Crowding
To understand why the EPFL discovery carries such massive implications for human health, one must examine the specific logistics of mitochondrial genetics. Unlike the rest of the cell, which stores its vast genome in the central nucleus, mitochondria possess their own distinct, circular DNA. This evolutionary remnant from when mitochondria were independent bacteria is absolutely critical for life. A single cell can contain hundreds to thousands of copies of mtDNA, which are packaged into dense protein-DNA clusters known as nucleoids.
These nucleoids must be spaced at remarkably precise intervals along the inner matrix of the mitochondrion. If the DNA clusters too closely together, the local concentration of transcribed proteins becomes unbalanced, leading to severe localized energy failures within the cell. Unregulated nucleoid distribution is a primary driver behind a host of metabolic and neurological conditions, including localized encephalopathy, severe liver failure, and the early progression of Parkinson’s and Alzheimer’s diseases.
For decades, the physical mechanism that kept these nucleoids neatly separated remained a total mystery. Researchers observed the even spacing but could not locate a transport mechanism or motor protein responsible for pushing the DNA clusters apart.
The Manley lab solved this by looking at the physical membrane itself. Using live-cell time-lapse imaging and super-resolution Stimulated Emission Depletion (STED) microscopy, they observed the mitochondria physically intervening. When calcium ions enter the mitochondrion, they trigger a sudden shift in membrane tension. The smooth outer and inner membranes rapidly pinch inward at regular intervals, forming a series of connected beads.
This sudden compartmentalization acts like an automated sorting machine. If a large, clumped nucleoid sits in the center of a newly formed "pearl," the physical tightening of the connecting membrane tubules forces the larger cluster to splinter. The smaller DNA fragments are squeezed into adjacent pearls. By the time the calcium levels normalize and the mitochondrion relaxes back into a smooth tube, the DNA has been successfully redistributed. The entire cycle executes in seconds.
Dismantling a 100-Year-Old Scientific Dogma
The idea that cellular membranes can transform into pearls on a string is not technically a new observation, but the interpretation of it represents a massive reversal of scientific dogma.
In 1915, pioneering cell biologists Margaret and Warren Lewis first observed mitochondrial pearling under early light microscopes. However, because their imaging methods required subjecting the cells to intense light and unnatural environments, the cells they observed were actively dying. Consequently, the scientific community classified membrane pearling as an irreversible pathological state—a final, terminal breakdown of the cell's structural integrity immediately preceding apoptosis (programmed cell death).
For the next century, this classification went largely unchallenged. Textbooks cemented the view that healthy organelles, axons, and plasma membranes maintained smooth, continuous, tubular structures. Any deviation into a beaded structure was automatically scored by researchers as an artifact of improper sample preparation or terminal cellular stress.
The optical revolution of the 2020s shattered this assumption. Standard confocal microscopy lacked the spatial resolution to see the inner workings of a 500-nanometer-wide mitochondrion, and traditional electron microscopy required freezing or fixing the cell, capturing only a static snapshot of a dead organism. The advent of ultra-fast live-cell imaging and zap-and-freeze electron microscopy allowed researchers to observe living cells at nanometer resolution in real-time.
By applying these non-destructive tools, biologists realized that pearling was not an irreversible death rattle. The cellular pearling process is a highly dynamic, reversible mechanism that healthy cells deploy constantly. The Lewises were not wrong in their 1915 observation; they were merely unaware that a healthy cell could reverse the process just as easily as it initiated it.
The Physics of the Beaded Membrane: Rayleigh-Plateau Instability
While the biological triggers for pearling differ across various cell types, the physical forces dictating the shape change are universal. The transition from a cylinder to a chain of pearls is driven by a phenomenon native to fluid dynamics known as the Rayleigh-Plateau instability.
First mathematically described in the 19th century by Joseph Plateau and Lord Rayleigh, this instability explains why a falling stream of water from a faucet eventually breaks up into individual droplets. A cylindrical column of liquid possesses a high surface area. Because surface tension naturally seeks to minimize surface area to reduce free energy, the cylinder is inherently unstable. Any slight perturbation in the fluid will cause the column to pinch inward and fragment into spheres, which hold the maximum volume for the minimum surface area.
Biological membranes operate under a more complex version of this physical law. Described by the Helfrich model of elastic membranes, cellular tubes are composed of lipid bilayers that resist bending. A mitochondrion or an axon remains tubular because the elastic bending energy of the membrane is strong enough to resist the surface tension pulling it toward a spherical shape.
The cellular pearling process occurs precisely when this delicate energy balance is tipped. If the internal osmotic pressure of an organelle increases, or if mechanical tension from the cell's cytoskeleton pulls on the membrane, the stretching energy suddenly overwhelms the membrane's bending stiffness. The membrane has no choice but to obey the laws of physics: it spontaneously curves into a series of pearls to alleviate the tension.
In the case of the EPFL discovery, calcium influx alters the osmotic pressure inside the mitochondria, deliberately initiating a Rayleigh-Plateau instability to force the nucleoids apart. Once the calcium is pumped back out, the pressure drops, the bending elasticity regains dominance, and the organelle smooths out. The cell is effectively hijacking the laws of fluid dynamics to do its internal housekeeping.
Axonal Pearling: Rewriting the Architecture of the Brain
The realization that membranes utilize controlled physical instability for survival extends far beyond the mitochondria. In 2024 and 2025, a research team led by Shigeki Watanabe at the Johns Hopkins University School of Medicine published data in Nature Neuroscience and Neuron that applied the pearling framework to the human brain.
Neurons communicate by sending electrical signals down long, thin cables called axons. Some human axons stretch up to a meter in length while remaining only about 100 nanometers thick. For a century, neuroscientists believed these axons were uniform, smooth cylinders, interrupted only by synaptic varicosities—the specialized bulges where neurotransmitters are stored and released to neighboring cells.
Watanabe’s team utilized zap-and-freeze electron microscopy to capture the activity of living mouse and human cortical tissue within milliseconds of an electrical stimulus. The resulting nanoscale imagery revealed that healthy, firing axons frequently adopt a pearls-on-a-string morphology.
These distinct, regularly spaced swellings—dubbed "non-synaptic varicosities"—measure about 250 nanometers across and are connected by ultra-thin membrane segments just 70 nanometers wide. Similar to the historical confusion surrounding mitochondria, axonal swelling had historically been documented exclusively as a marker of traumatic brain injury, oxygen deprivation, or neurodegeneration. Watanabe’s data proved that these pearl-like structures appear naturally in healthy neural tissue.
In the brain, the cellular pearling process appears to be tied to ultrafast endocytosis, a rapid membrane retrieval system. When a neuron fires rapidly, it must quickly recycle the synaptic vesicles used to transmit the signal. The physical membrane of the axon temporarily pearls to compartmentalize specific regions, isolating the biochemical machinery required to pull the membrane back inside the cell quickly and efficiently.
If an axon loses the ability to reverse this pearled state, the transmission of electrical signals is severely bottlenecked, mimicking the exact pathology seen in multiple sclerosis and severe concussive trauma. The brain depends on the transient, reversible nature of these structures to manage the massive membrane turnover required for learning and memory.
Platelet Biogenesis: Precision Engineering in the Bloodstream
The utility of membrane pearling scales up to even larger physiological systems. In June 2025, researchers mapping the circulatory system uncovered how the human body utilizes pearling to dictate the exact physical size of blood platelets.
Platelets, the cells responsible for blood clotting and wound healing, are not born as independent entities. They are physically ripped away from massive precursor cells in the bone marrow called megakaryocytes. These giant cells extend long, tubular membrane protrusions called proplatelets through the walls of bone marrow blood vessels directly into the flowing bloodstream.
For decades, hematologists struggled to explain how the megakaryocyte ensures that every platelet breaking off into the blood is exactly the right size—roughly 2 to 3 micrometers in diameter. If platelets are too large, they can trigger deadly thrombotic events, causing strokes or heart attacks. If they are too small, they fail to aggregate effectively, leading to uncontrolled hemorrhage.
The answer lies in mechanically induced membrane pearling. As the megakaryocyte extends its cylindrical proplatelet tube into the blood vessel, the sheer force of the flowing blood applies immense mechanical tension to the membrane. This hydrodynamic stress alters the tension-to-elasticity ratio of the lipid bilayer, intentionally triggering a Rayleigh-Plateau instability.
The long proplatelet tube spontaneously converts into a string of uniform pearls. Because the physical parameters of the membrane—its lipid composition and stiffness—are highly regulated by the cell, the mathematical wavelength of the instability is fixed. This ensures that every resulting pearl is identical in volume. As the sheer flow of the blood continues to pull on the string, the thin interconnecting tubules eventually snap, releasing a perfectly calibrated payload of identically sized platelets into the circulation.
In this context, the pearling instability acts as a master mechanobiological regulator. By exploiting external fluid dynamics, the megakaryocyte avoids the need for complex, energy-heavy enzymatic cleavage machinery, relying instead on pure physics to mass-produce the cellular components necessary to stop human bleeding.
The Role of the Cytoskeleton: Actin and BAR Domain Proteins
While external forces like blood flow or internal triggers like calcium-induced osmotic shifts can initiate pearling, the cell often requires a physical scaffold to control the process. Unregulated Rayleigh-Plateau instability would quickly tear a cell membrane apart, transforming the entire organism into a collection of disjointed lipid droplets. To harness the instability, cells deploy specialized architectural proteins.
Chief among these regulatory networks is the actin cortex, a dense meshwork of protein filaments that lies just beneath the plasma membrane. Research utilizing cryo-electron tomography on various cell types, including Dictyostelium and mammalian fibroblasts, has shown that the actin cortex acts as a shock absorber. It suppresses spontaneous pearling by providing a rigid structural backing that artificially increases the membrane’s resistance to bending.
When a cell needs to pearl—whether to divide, move, or compartmentalize—it must first locally dismantle this actin support. By locally depolymerizing the actin filaments, the cell removes the structural backing, allowing the naked lipid bilayer to succumb to surface tension and pearl. The state remains stable as long as the actin is actively prevented from repolymerizing. Once the task is complete, the cell quickly reassembles the actin scaffold, forcing the pearls to merge back into a flat or tubular membrane.
In addition to actin manipulation, cells utilize specialized curvature-inducing proteins, most notably BAR (Bin/Amphiphysin/Rvs) domain proteins. These crescent-shaped proteins physically bind to the lipid bilayer, mechanically bending it. Depending on their concentration and orientation, adsorbed curved proteins can either stabilize a membrane tube or actively drive it toward pearling. When BAR domain proteins recruit localized actin polymerization, the growing actin filaments create an inward constrictive force, squeezing the membrane tube until it pearls and eventually undergoes fission, splitting into separate vesicles. This localized, protein-driven pearling is the fundamental physical mechanism driving endocytosis—the process by which cells swallow nutrients, viruses, and signaling molecules from their external environment.
Synthetic Biology and the Origins of Life
Because the cellular pearling process is fundamentally driven by the physical properties of lipid membranes rather than complex DNA-encoded protein machinery, it provides a compelling window into the origins of cellular life on Earth.
Long before the evolution of complex divisome proteins like FtsZ or actin-myosin contractile rings, the first primitive cells (protocells) needed a way to grow and divide to pass on their genetic material. Origin-of-life researchers heavily focus on fatty acid vesicles as the most prebiotically plausible models for these early membranes, as fatty acids can be synthesized through simple, non-biological chemical reactions and spontaneously self-assemble into hollow spheres.
Recent experiments in membrane mimetic chemistry have successfully reproduced cellular division in the laboratory using nothing but light and basic physics. Researchers illuminated filamentous fatty acid vesicles with ultraviolet light in the presence of simple thiols. The resulting light-driven chemical reaction altered the surface tension of the vesicle membrane. Within seconds, the continuous fatty acid tube underwent a massive pearling instability, transforming into a chain of distinct vesicles.
Because these early membranes lacked the actin cortex or stabilizing proteins of modern cells, the thin tubules connecting the pearls eventually snapped under ambient thermal energy, resulting in independent, perfectly divided daughter cells. This demonstrates that the earliest form of cellular reproduction may not have been a biological innovation, but rather a passive surrender to membrane physics.
Today, synthetic biologists are harnessing this ancient mechanism to design advanced artificial cells. By tuning the lipid composition of lab-grown liposomes and incorporating light-sensitive or pH-sensitive molecules, engineers can force synthetic membranes to pearl on command. This allows for the creation of smart drug-delivery vehicles that circulate safely in the bloodstream as stable tubes but undergo pearling and fragmentation to release highly localized payloads of chemotherapy drugs only when exposed to the specific acidic microenvironment of a tumor.
Clinical Ramifications and Neurodegenerative Disease
The revelation that pearling is a vital physiological process rather than a mere artifact of cell death completely recontextualizes how pharmaceutical companies are approaching metabolic and neurodegenerative diseases. If pearling is required to maintain internal cellular order, then the failure of a cell to pearl—or its inability to escape a pearled state—represents a distinct class of mechanical disease.
The EPFL data connecting mitochondrial pearling to DNA nucleoid distribution offers an immediate application. In diseases like Alzheimer's and Parkinson's, one of the earliest observable cellular defects is mitochondrial dysfunction resulting from clumped, disorganized mtDNA. Without the proper spacing of these genetic blueprints, the mitochondria fail to produce the localized proteins needed to maintain the electron transport chain, leading to a massive spike in toxic reactive oxygen species that eventually kill the neuron.
If this mtDNA clumping is the direct result of a mechanical failure in the cellular pearling process, it opens an entirely new therapeutic vector. Currently, treatments for mitochondrial dysfunction focus on metabolic supplements or antioxidants intended to neutralize the resulting toxic byproducts. However, if researchers can identify the specific genetic or pharmacological triggers that initiate mitochondrial pearling—such as modulating local calcium channels or targeting the inner membrane structural proteins—they could theoretically force the mitochondria to undergo a pearling cycle. This artificial induction could break up the nucleoid clumps and restore healthy energy production before the neuron suffers permanent oxidative damage.
Conversely, in the context of Shigeki Watanabe's axonal research, pathology often arises when the membrane becomes trapped in the pearled configuration. When an axon undergoes severe concussive trauma, the physical shockwave causes a massive, unregulated influx of calcium and a sudden disruption of the microtubule scaffolding. The axon immediately pearls, but the sheer volume of the physical disruption destroys the cell's ability to pump the calcium back out and repolymerize the internal skeleton. The axon becomes frozen in a pearls-on-a-string morphology, permanently halting electrical transmission and leading to the immediate cognitive deficits associated with severe traumatic brain injury.
Understanding the precise biophysical thresholds of the Rayleigh-Plateau instability in human tissue allows researchers to develop membrane-stabilizing drugs. By administering compounds that artificially increase the bending elasticity of the axonal membrane immediately following a concussive event, emergency room physicians could mathematically prevent the tension threshold from being breached, stopping axonal pearling before it leads to permanent nerve death.
The Next Decade of Cellular Architecture
The discovery timeline spanning 2024 to May 2026 has irrevocably altered the landscape of biophysics and cellular imaging. As scientists continue to deploy sub-diffraction-limit microscopy and high-speed cryo-electron tomography, the static textbook illustrations of cellular organelles are being replaced by dynamic, physics-driven models.
Moving forward, the primary focus of cellular biophysics will center on the precise regulatory mechanisms that dictate when a pearl remains part of a connected chain and when it undergoes final fission. The physical transition from a connected pearl to a fully separated vesicle requires the complete severing of the lipid bilayer—a process that is highly energetically unfavorable. While fluid dynamics can drive the membrane into a pearled shape, it is currently debated whether pure physics is sufficient to snap the connecting tubule in healthy intracellular environments, or if specialized scission proteins like the dynamin-like protein Drp1 are universally required to finish the job.
Preliminary data suggests that manipulating these scission proteins can trap organelles in transient states, offering an unprecedented opportunity to map the biochemical gradients inside individual pearls. Researchers are actively investigating how the isolation of specific enzymes inside a mitochondrial pearl might temporarily alter the local metabolic output of that specific region of the cell.
Furthermore, the expansion of three-dimensional cell culturing is revealing new modalities of cell division. When cells divide in traditional, flat, two-dimensional petri dishes, they round up into spheres. However, recent studies observing cell division in native-like 3D microenvironments reveal that cells exhibit long, extended protrusions that undergo spontaneous, quasiperiodic membrane pearling. This suggests that the spatial constraints of real biological tissues rely heavily on pearling instabilities to guide the orientation and mechanical separation of dividing cells—a factor that has been largely ignored in decades of 2D laboratory research.
The convergence of fluid dynamics, super-resolution microscopy, and clinical pathology has unveiled a hidden mechanical language operating inside our bodies. The cells keeping us alive are not rigid machines built of static parts; they are highly malleable, fluid ecosystems that constantly negotiate with the laws of physics. By learning to read and eventually dictate this mechanical language, the next generation of medical science will be uniquely positioned to repair the fundamental architecture of human life, one microscopic pearl at a time.
Reference:
- https://www.sciencedaily.com/releases/2026/04/260413043133.htm
- https://scitechdaily.com/scientists-just-discovered-the-hidden-trick-that-keeps-your-cells-alive/
- https://www.molbiolcell.org/doi/10.1091/mbc.E25-06-0302
- https://www.youtube.com/watch?v=mjS2aIEJMQ0
- https://www.biorxiv.org/content/10.1101/2024.12.21.629509v1.full-text
- https://scitechdaily.com/scientists-debunk-100-year-old-belief-about-brain-cells-rewriting-textbooks/
- https://www.researchgate.net/publication/393023119_Tube_into_pearls_A_membrane-driven_pearling_instability_shapes_platelet_biogenesis
- https://pubs.acs.org/doi/10.1021/jacs.1c03436
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4026785/
- https://cordis.europa.eu/project/id/273192/reporting/it
- https://pubmed.ncbi.nlm.nih.gov/26716426/
- https://www.researchgate.net/publication/266204220_Pearling_instability_of_membrane_tubes_driven_by_curved_proteins_and_actin_polymerization
- https://www.researchgate.net/publication/335881864_Protrusion_membrane_pearling_emerges_during_3D_cell_division