Medical intervention for severe cognitive decline has historically operated within a frustrating paradigm: physicians can observe the structural deterioration of the mind, track the erosion of memory, and measure the accumulation of toxic proteins, but they possess severely limited tools to stop the underlying destruction. By the time a patient presents with clinical memory loss, the architecture of the hippocampus has often been subjected to years of relentless, microscopic damage.
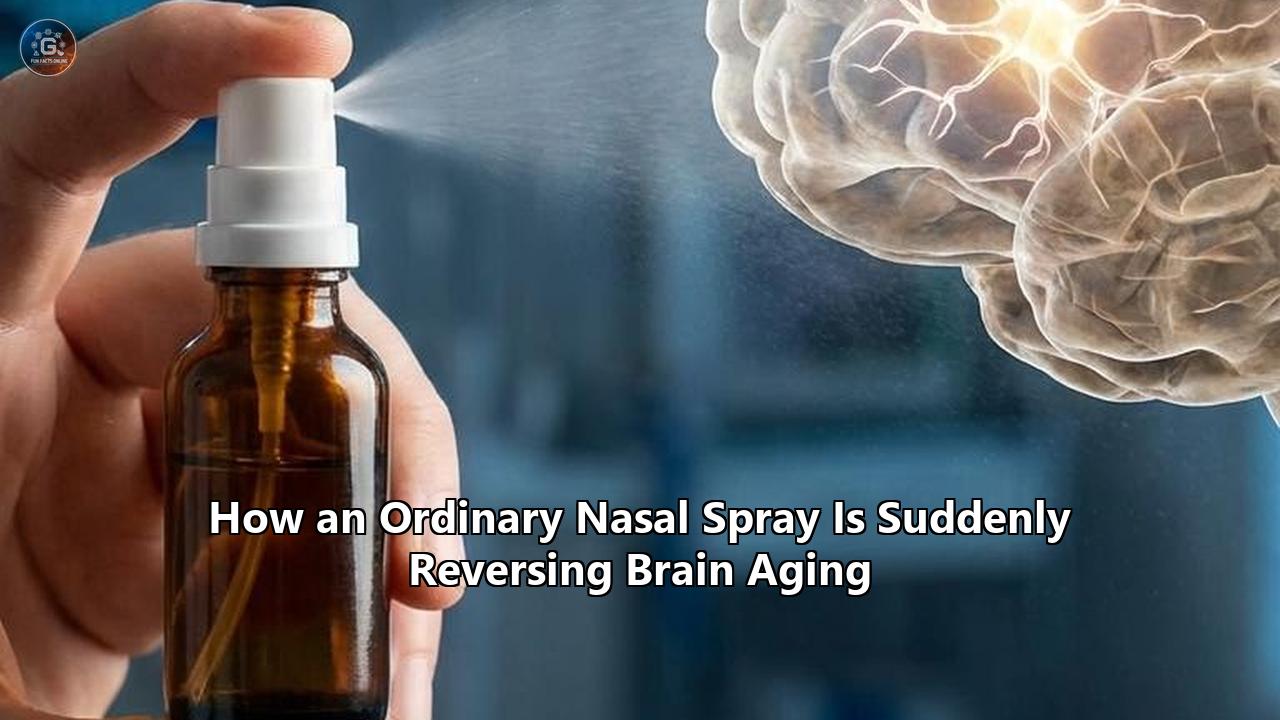
In mid-April 2026, researchers at the Texas A&M University Naresh K. Vashisht College of Medicine published a landmark study in the Journal of Extracellular Vesicles that effectively upends this dynamic. Led by Dr. Ashok Shetty, distinguished professor and associate director at the Institute for Regenerative Medicine, the research team successfully reversed age-related brain inflammation and memory decline in preclinical models using a non-invasive, two-dose therapy. The delivery mechanism was remarkably straightforward: a targeted nasal mist.
The findings arrive at a critical juncture for global health infrastructure. For researchers tracking the efficacy of this nasal spray brain aging models in the laboratory demonstrate that cognitive decline is not an inevitable, one-way street. The intervention rapidly cleared neuroinflammation, recharged the cellular power plants within neurons, and restored memory function, with behavioral improvements materializing within weeks and persisting for months.
To grasp the magnitude of this development, it is necessary to examine the specific biological failures that drive dementia, why traditional pharmacology has repeatedly failed to intercept them, and how an innovative biological courier system is rewriting the rules of neurological care.
Unmasking the Crisis of Neuroinflammaging
The underlying problem revealed by the Texas A&M study centers on a physiological state known as "neuroinflammaging." For decades, the medical consensus held that brain aging was a passive consequence of cellular wear and tear—a gradual slowing of metabolic processes across the central nervous system. Modern neurobiology paints a far more violent picture. Aging does not happen quietly; it is driven by a chronic, slow-burning inflammatory fire that takes root deep within the brain, particularly targeting the hippocampus, the neurological center responsible for learning, spatial navigation, and memory consolidation.
The primary instigators of this biological fire are microglia. In a healthy, youthful brain, microglia function as the resident immune system and diligent caretakers of the neural environment. They continuously patrol the central nervous system, identifying and clearing away dead cells, metabolic debris, and misfolded proteins. They prune unused synapses to keep neural networks efficient.
However, as mammalian brains age, these once-protective immune cells undergo a severe structural and functional mutation. Driven by accumulated oxidative stress and genetic transcription errors, the microglia become hyperactive and perpetually agitated. Instead of methodically clearing debris, they enter a state of constant high alert, releasing a continuous stream of inflammatory signaling proteins known as cytokines. The microglial network effectively turns against the tissue it was designed to protect, transforming the delicate environment of the hippocampus into a highly toxic, inflamed battleground.
This localized inflammation destroys the brain's ability to maintain healthy synaptic connections. As Dr. Shetty outlined following the publication of the data, the slow burn of neuroinflammaging creates a persistent neurological fog that makes it difficult for aging individuals to process complex thoughts, form new memories, or adapt to novel environments. It is the upstream catalyst that vastly increases the risk of severe disorders, including Alzheimer's disease and other forms of progressive dementia.
The Fortress of the Mind: Blood-Brain Barrier Obstacles
Identifying hyperactive microglia as the root cause of cognitive decline is only half the diagnostic equation; neutralizing them presents a formidable logistical challenge. The central nervous system is entirely encased in an impenetrable vascular fortress known as the blood-brain barrier (BBB). This highly selective, semipermeable border of tightly packed endothelial cells prevents circulating blood pathogens, toxins, and fluctuations in systemic immune responses from crossing into the extracellular fluid of the brain and spinal cord.
While the blood-brain barrier is highly effective at keeping dangerous pathogens out, it is equally effective at blocking therapeutic agents. The vast majority of traditional anti-inflammatory drugs, whether injected intravenously or administered orally, cannot penetrate this endothelial wall in molecular concentrations high enough to alter the microglial environment. The molecules are simply too large, or their lipid solubility is too low, to pass through the tight junctions of the barrier.
When pharmaceutical developers have attempted to physically bypass the blood-brain barrier using invasive surgical procedures—such as direct cerebrospinal fluid injections, intrathecal pumps, or cranial drilling—the clinical risks often negate the potential benefits. Invasive neural access introduces the threat of severe infection, localized tissue trauma, and immense patient discomfort. The underlying premise of reversing cognitive decline with a nasal spray brain aging intervention hinges on bypassing the vascular system entirely, avoiding the surgical risks that have historically bottlenecked neurological therapeutics.
The Pathological Misfire: DNA Leaks and Cytokine Storms
To engineer a precise solution, the Texas A&M team first had to map the exact molecular pathways that trigger the microglial revolt. Their research isolated two primary culprits responsible for turning the hippocampus into an inflammatory zone: the NLRP3 inflammasome and the cGAS-STING signaling pathway.
The NLRP3 inflammasome is a multiprotein complex expressed within myeloid cells, including microglia. Under normal conditions, it lies dormant. However, when triggered by cellular stress signals common in older brains—such as the accumulation of amyloid plaques or high levels of reactive oxygen species—the inflammasome activates. Once engaged, it catalyzes the maturation and rapid release of highly potent pro-inflammatory cytokines, specifically Interleukin-1 beta (IL-1β) and Tumor Necrosis Factor-alpha (TNF-α). These cytokines flood the local tissue, actively degrading the myelin sheaths that insulate neural pathways.
Simultaneously, the cGAS-STING pathway begins to misfire. Originally evolved as an ancient cellular defense mechanism to detect the presence of stray DNA from invading viruses, this pathway becomes a liability in aging cells. As neurons age, their internal structures degrade. Specifically, the mitochondria—the cellular power plants—begin to break down and leak their own mitochondrial DNA into the cellular cytoplasm. The cGAS-STING pathway detects this leaked internal DNA, mistakenly identifies it as a foreign viral infection, and triggers a massive, localized immune response.
The resulting cytokine storms starve the surrounding neurons. Because neural cells require immense amounts of adenosine triphosphate (ATP) to maintain their electrical resting potentials and fire action potentials across synapses, they are highly sensitive to energy disruptions. Under constant inflammatory assault, mitochondrial function collapses. Oxidative stress spikes, energy production plummets, and the neurons lose the electrical capacity to form or retrieve memories. The cellular engine stalls.
The Economic Weight of the Coming Cognitive Cliff
The necessity for a viable intervention transcends cellular biology; it is a macroeconomic imperative. Global health systems face a demographic crisis that threatens to overwhelm eldercare infrastructure within the next two decades.
In the United States alone, the prevalence of dementia is tracking toward catastrophic levels. Driven by the aging of the baby boomer generation and increased baseline life expectancies, cases of dementia are projected to climb from a steady baseline to nearly 13 million individuals by 2050. The economic burden of neurodegenerative diseases, characterized by the need for prolonged, high-intensity, round-the-clock caregiving, already consumes hundreds of billions of dollars annually. Without an intervention that halts or reverses the physiological progression of the disease, the financial and social costs will become mathematically unsustainable for public health programs.
"The trend signals a pressing need for policies and innovative interventions that can minimize both the risk and severity of neurodegenerative disorders like dementia," Dr. Shetty stated, addressing the broader context of the research.
Historically, clinical efforts have focused almost exclusively on managing late-stage symptoms. Billions of dollars in research funding have been channeled into monoclonal antibodies designed to clear amyloid-beta plaques from the brain long after the structural damage to the hippocampus has occurred. The Texas A&M approach represents a fundamental pivot: targeting the upstream inflammation and mitochondrial collapse before it triggers irreversible neuronal death.
The Engineered Solution: Harnessing Extracellular Vesicles
Faced with the twin hurdles of an impenetrable blood-brain barrier and the complex molecular biology of hyperactive immune cells, Dr. Shetty's team, including senior research scientists Dr. Madhu Leelavathi Narayana and Dr. Maheedhar Kodali, engineered a highly specialized biological delivery mechanism. They discarded traditional synthetic pharmacology in favor of the body's own communication networks, utilizing extracellular vesicles (EVs).
Extracellular vesicles are naturally occurring, microscopic lipid-bound particles that cells use to transmit information to one another across distances. Measuring roughly 130 nanometers in diameter, these vesicles function as secure biological couriers, packaging and transporting proteins, lipids, and genetic instructions while shielding their cargo from extracellular degradation.
To source these therapeutic couriers, the research team utilized human induced pluripotent stem cell-generated neural stem cells (hiPSC-NSCs). Pluripotent stem cells are uniquely valuable because they can be programmed in the laboratory to develop into virtually any cell type in the human body. By guiding these cells to become neural stem cells, the researchers cultivated a population of cells that naturally secrete extracellular vesicles perfectly calibrated for integration into human brain tissue.
The Genetic Cargo: Silencing Inflammation with MicroRNA
The critical innovation of the Texas A&M study lies not just in the vesicles themselves, but in the specific cargo they carry. The researchers loaded the EVs with isolated microRNAs—tiny fragments of non-coding RNA that function as epigenetic dimmers, capable of regulating and suppressing the expression of targeted genes.
Specifically, the vesicles were enriched with miR-30e-3p and miR-181a-5p, microRNAs clinically recognized for their potent anti-inflammatory properties. Unlike traditional small-molecule drugs that attempt to block the receptors of inflammatory cytokines after they have already been released, microRNAs intervene at the genetic level. When introduced into a cell, they bind directly to specific messenger RNA (mRNA) sequences, preventing the cell's ribosomes from translating those sequences into proteins.
"MicroRNAs act like master regulators," explained Dr. Narayana. "They help modulate and regulate many gene and signaling pathways in the brain."
By packing the extracellular vesicles with these specific genetic regulators, the team created a therapy designed to intercept the neuroinflammaging process at its source. The goal was to deliver the microRNAs directly into the hyperactive microglia, genetically silencing the instructions that command the NLRP3 inflammasome and cGAS-STING pathways to produce toxic cytokines.
The Anatomical Backdoor: Delivering Therapy Through the Olfactory System
Having engineered the therapeutic payload, the researchers faced the final logistical hurdle: bypassing the blood-brain barrier to deliver the vesicles into the hippocampus. Intravenous administration would result in the vesicles circulating harmlessly in the bloodstream, entirely blocked from the central nervous system.
To solve this, the team utilized a physiological backdoor: the olfactory route. By formulating the hiPSC-NSC-EV treatment into a fine mist, the researchers exploited the direct anatomical connection between the nasal cavity and the brain.
The roof of the human nasal cavity contains the olfactory epithelium, a specialized tissue packed with olfactory receptor neurons. These are the only central nervous system neurons that extend directly into the external environment. When the therapeutic mist is administered, the microscopic vesicles are absorbed by the nasal mucosa and travel directly along the olfactory and trigeminal nerve fibers. This specific neural pathway bypasses the circulatory system and the blood-brain barrier entirely, allowing the extracellular vesicles to travel uninterrupted from the nasal cavity straight into the brain tissue, pooling heavily in the memory centers of the hippocampus.
Dr. Kodali emphasized the clinical superiority of this delivery architecture: "The mode of delivery is one of the most exciting aspects of our approach. Intranasal delivery allows us to reach, and treat, the brain directly without invasive procedures."
The implications for patient care are immense. A therapy that can be administered via a nasal spray eliminates the need for hospital admission, sterile surgical theaters, and complex intravenous infusions. By the time clinical trials validate the nasal spray brain aging could be fundamentally redefined across the medical community, shifting cognitive care from specialized neurology wards directly into outpatient clinics and primary care settings.
Restoring the Spark: Mitochondrial Recovery and Behavioral Success
The true measure of the therapy's success occurred once the vesicles reached their target. Upon entering the hippocampus, the extracellular vesicles were readily absorbed by the localized microglia. The microRNA cargo (miR-30e-3p and miR-181a-5p) deployed, immediately binding to the target mRNA.
The biological results were stark and rapid. The genetic instructions for the NLRP3 inflammasome and cGAS-STING pathways were silenced. The localized production of IL-1β and TNF-α plummeted. With the cytokine storm halted, the surrounding neural environment stabilized.
Freed from the constant barrage of oxidative stress, the neurons initiated their own cellular repair mechanisms. The treatment physically recharged the neuronal mitochondria—the power plants of the cells. As ATP production normalized, the neurons regained their capacity to fire action potentials and maintain synaptic integrity.
"We are giving neurons their spark back by reducing oxidative stress and reactivating the brain's mitochondria," Dr. Narayana observed, tracking the cellular recovery in real time.
This microscopic metabolic recovery translated into profound behavioral restoration. In rigorous laboratory testing, aging animal models treated with just two doses of the nasal spray—spaced two weeks apart—underwent extensive cognitive evaluation. The researchers utilized object recognition tests and spatial memory assays to measure neural function.
The models that received the therapeutic mist displayed a dramatic resurgence in cognitive sharpness. They demonstrated a clear, restored ability to recognize familiar objects, detect entirely novel items placed in their environment, and adapt to sudden spatial changes. These are precise cognitive metrics that typically collapse under the weight of neurodegeneration. The treated models responded to environmental stimuli with the behavioral acuity of much younger subjects.
Crucially, the treatment proved universally effective across the test population. Unlike many experimental neurological interventions that show varying degrees of efficacy based on biological sex, the EV nasal spray yielded identical, positive treatment outcomes in both male and female subjects.
"We are seeing the brain's own repair systems switch on, healing inflammation and restoring itself," Dr. Shetty concluded. The most highly anticipated metric of the study was durability: the cognitive improvements did not fade rapidly. Following the two-dose regimen, the behavioral benefits persisted for months, indicating structural healing rather than a temporary chemical stimulation.
Scaling the Therapy: Patents, Trials, and Future Applications
The transition from successful animal models to approved human therapeutics is a notoriously difficult gauntlet, heavily scrutinized by regulatory agencies. However, the foundational mechanics of the Texas A&M therapy offer distinct advantages over synthetic pharmacology. Because the intervention relies on naturally occurring biological messengers—human-derived extracellular vesicles—rather than synthetic foreign compounds, the risk of severe immunogenic rejection or systemic toxicity is sharply reduced. Furthermore, the non-invasive nature of the olfactory delivery system significantly accelerates the pathway to human safety trials, as the risk profile of an intranasal mist is exponentially lower than that of surgical implantation or heavy systemic dosing.
Supported by collaborative resources from the National Institute on Aging (NIA), Dr. Shetty and the research team have officially filed a United States patent for the EV therapy. "Our partnership with the NIA is very important," Dr. Shetty noted. "This kind of work requires resources and the right people to tackle problems and develop solutions that could change lives."
The immediate next phase of development requires scaling the laboratory production of hiPSC-NSC-derived extracellular vesicles to pharmaceutical grades. Manufacturing millions of standardized, quality-controlled vesicles loaded with precise microRNA concentrations is a complex biomanufacturing challenge that must be solved before initiating Phase 1 human clinical trials.
Beyond treating age-related memory decline, the underlying technology offers expansive therapeutic horizons. The ability to non-invasively regulate microglial activity and suppress neuroinflammation provides a versatile tool for treating a wide array of neurological crises. Researchers theorize that similar EV-based nasal therapies could be adapted to aid stroke survivors, helping to clear acute inflammation and rebuild lost neural function in the immediate aftermath of a cerebral infarction. The technology also holds immense promise for mitigating the chronic inflammation associated with traumatic brain injuries (TBI) and severe concussions.
Medical leaders are beginning to shift their operational frameworks away from merely managing the final, devastating stages of cognitive collapse. The emerging clinical goal is "successful aging"—a sustained physiological state where the brain remains physically resilient, the memory center remains intact, and individuals stay socially engaged and intellectually connected well into their later decades.
As public health officials review the implications of the nasal spray brain aging demographics may finally encounter a scalable solution. By identifying the specific molecular fires that destroy the mind and engineering a precise, non-invasive method to extinguish them, researchers have demonstrated that the aging brain retains a profound capacity for self-repair. The neurons simply require the right biological signals, delivered directly to their doorstep, to reignite their spark.
Reference:
- https://www.earth.com/news/new-nasal-spray-reverses-brain-aging-in-breakthrough-study/
- https://stories.tamu.edu/news/2026/04/14/scientists-reverse-brain-aging-with-a-nasal-spray/
- https://neurosciencenews.com/nasal-spray-reverse-brain-aging-30519/
- https://www.miragenews.com/nasal-spray-reverses-brain-aging-scientists-find-1655162/
- https://www.academicjobs.com/us/research-publication-news/texas-aandm-nasal-spray-reverses-brain-aging-or-academicjobs-13372
- https://usaherald.com/nasal-spray-reverses-brain-aging-in-stunning-study-restores-memory-and-slashes-inflammation/
- https://www.techexplorist.com/nasal-spray-reverse-brain-aging/102701/
- https://podtail.se/podcast/dr-howard-smith-reports/instinct-cordless-hair-clippers-overheat/
- https://www.iflscience.com/nasal-spray-could-reverse-brain-aging-by-lowering-inflammation-and-re-energizing-brain-cells-83226