The world is quietly slipping into a new, terrifying epoch: the post-antibiotic era. For nearly a century, we have enjoyed the luxury of treating previously fatal bacterial infections with a simple pill or an intravenous drip. A scratched knee, a routine surgery, or a common case of strep throat were stripped of their lethal potential thanks to the miracle of antibiotics. But that golden age is rapidly drawing to a close. Bacteria, the oldest and most adaptable organisms on Earth, are fighting back—and they are winning.
According to a sobering 2024 study published in The Lancet, more than 39 million people worldwide could die from antibiotic-resistant infections between 2025 and 2050, equating to roughly three deaths every single minute. In the fall of 2025, the World Health Organization (WHO) issued its Global Antimicrobial Resistance and Use Surveillance System (GLASS) report, revealing that one in six laboratory-confirmed bacterial infections are now resistant to standard treatments. Between 2018 and 2023 alone, resistance in targeted pathogen-antibiotic combinations surged by an average of 5% to 15% annually. Meanwhile, the U.S. Centers for Disease Control and Prevention (CDC) recently reported a staggering 460% increase in cases of NDM-CRE—a highly lethal, drug-resistant "superbug" that evades almost all known medical interventions.
Humanity has inadvertently engineered its own biological nightmare through the overuse and misuse of antibiotics in medicine and agriculture. As pharmaceutical pipelines run dry and multidrug-resistant pathogens evolve at breakneck speed, modern medicine finds itself standing at the edge of a precipice.
Yet, there is a glimmer of hope. To fight an ancient, rapidly mutating enemy, scientists are enlisting an even older, more relentless predator. Enter the bacteriophage: a microscopic, spider-like virus that exists for one single, unwavering purpose—to hunt, infect, and obliterate bacteria.
Meet the Phage: Nature’s Perfect Assassin
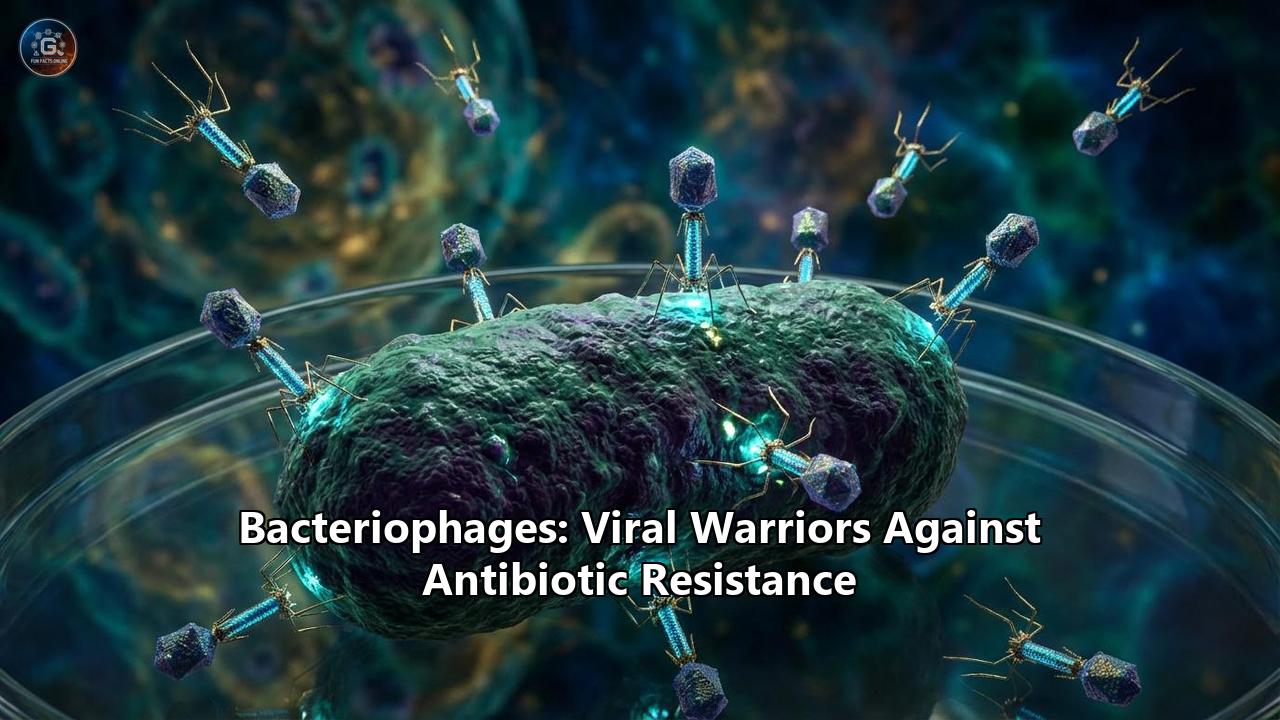
Bacteriophages, or simply "phages," are viruses that exclusively infect bacteria. You are covered in them right now. They are in the water you drink, the soil you walk on, and deep within your gut microbiome. With an estimated ten nonillion ($10^{31}$) phages on the planet, they outnumber every other biological entity on Earth combined. Where there are bacteria, there are phages preying upon them, locked in an invisible, billion-year-old evolutionary arms race.
Structurally, a typical bacteriophage looks like a microscopic alien spacecraft. It features an icosahedral (20-sided) protein head, or capsid, which houses tightly coiled genetic material (DNA or RNA). This head sits atop a hollow protein tail, complete with spindly tail fibers that act like landing gear. But these fibers are not just for touching down; they are highly sophisticated chemical sensors.
Phages are the ultimate precision weapons. Unlike broad-spectrum antibiotics, which act like biological napalm—wiping out both pathogenic bacteria and the beneficial microbes in your body—phages are generally highly specific. A phage that targets Escherichia coli will not harm Staphylococcus aureus, nor will it touch human cells. It is this extraordinary specificity that makes them the most promising candidates to bypass the antibiotic resistance crisis.
When a phage encounters its specific bacterial target, a ruthless biological hijacking occurs. This is known as the lytic cycle:
- Attachment: The phage uses its tail fibers to bind to specific receptors on the bacterial cell wall. If the receptors don't match perfectly, the phage moves on.
- Penetration: The phage acts like a microscopic syringe, driving its tail tube through the bacterial membrane and injecting its genetic material into the host.
- Hijacking: The viral DNA immediately takes command of the bacterium's internal machinery. The bacterium stops its normal functions and is forced into becoming a phage-manufacturing factory.
- Assembly: Hundreds, sometimes thousands, of new phage components are synthesized and assembled inside the dying bacterial cell.
- Lysis: The newly formed phages produce an enzyme called endolysin, which compromises the bacterial cell wall from the inside out. The bacterium explodes (lyses), releasing the new phages into the environment to hunt down more bacteria.
This localized "auto-dosing" phenomenon means that the medicine actually multiplies at the site of the infection, growing in numbers exactly where it is needed, and then naturally clearing from the body once the bacterial food source is eliminated.
A Forgotten History: From the Ganges to the Cold War
While the Western world is only just waking up to the potential of phage therapy, the concept is far from new. Long before antibiotics were discovered, phages were being observed and utilized, albeit without a full understanding of what they were.
In 1896, British bacteriologist Ernest Hankin reported that the waters of the Ganges and Yamuna rivers in India contained a mysterious, heat-labile substance capable of killing the cholera bacteria, drastically limiting the spread of outbreaks. Two decades later, during the First World War, British bacteriologist Frederick Twort and French-Canadian microbiologist Félix d'Hérelle independently isolated these bacterial killers. It was d'Hérelle who named them bacteriophages—from the word "bacteria" and the Greek phagein, meaning "to devour".
D'Hérelle was a maverick. In 1919, he successfully used phages to treat an outbreak of dysentery in a Parisian hospital, effectively conducting the first clinical application of phage therapy. As enthusiasm grew, commercial phage laboratories sprung up across Europe and the United States in the 1920s and 1930s.
However, the science of the time was rudimentary. Phages were not fully understood; they were prescribed for viral infections (which they cannot cure), mishandled, and improperly purified. The results were inconsistent. When Alexander Fleming discovered penicillin in 1928, and it was successfully mass-produced in the 1940s, antibiotics stole the spotlight. Antibiotics were stable, easy to patent, easy to manufacture, and they worked against a broad spectrum of diseases without needing to identify the exact bacterial strain. The West abandoned phages almost overnight.
But behind the Iron Curtain, the story was vastly different. George Eliava, a Georgian scientist who had trained with d'Hérelle in Paris, established the Eliava Institute of Bacteriophage, Microbiology, and Virology in Tbilisi, Georgia, in 1923. Because the Soviet Union lacked access to Western antibiotics during the Cold War, they heavily funded the Eliava Institute. Phage therapy became a standard part of Soviet medicine. Soldiers carried phage cocktails in their field kits to treat wound infections and dysentery. To this day, in Georgia, Russia, and parts of Eastern Europe, phage preparations are sold over the counter in pharmacies.
The Clinical Renaissance: Modern Miracles and Compassionate Use
Fast forward to the 21st century. The antibiotic miracle has faltered, and Western scientists are suddenly looking eastward, dusting off old Soviet research, and modernizing the science of phage therapy using next-generation genomic sequencing. Over the last decade, high-profile medical cases have thrust phages back into the limelight.
Perhaps the most famous modern catalyst for phage therapy is the case of Dr. Tom Patterson in 2016. While vacationing in Egypt, Patterson was infected by a multidrug-resistant strain of Acinetobacter baumannii. After returning to the United States, his condition rapidly deteriorated. His organs were failing, he slipped into a coma, and no available antibiotics could stop the superbug. Desperate, his wife, infectious disease epidemiologist Dr. Steffanie Strathdee, initiated a global hunt for phages. Researchers from the U.S. Navy and Texas A&M University successfully isolated, purified, and engineered a "cocktail" of specific phages. Administered under the U.S. Food and Drug Administration's (FDA) emergency Investigational New Drug (eIND) protocol, the phages were pumped directly into Patterson's bloodstream. He woke from his coma shortly after and made a full recovery.
This watershed moment triggered a cascade of similar compassionate-use cases. Today, hundreds of patients suffering from multidrug-resistant bacterial infections are treated yearly under expanded access programs.
- Cystic Fibrosis: Patients with cystic fibrosis often develop chronic, life-threatening lung infections from superbugs like Mycobacterium abscessus and Pseudomonas aeruginosa. In a recent case, a patient infected with drug-resistant M. abscessus was treated with genetically engineered phages (a cocktail of BPsΔ33HTH_HRM10 and D29_HRMGD40). The treatment drastically reduced the bacterial load, clearing the way for a successful, life-saving lung transplant.
- Periprosthetic Joint Infections (PJI): When joint replacements (like knees or hips) become infected, bacteria often form protective shields called biofilms, rendering antibiotics useless and normally requiring devastating surgical amputations. Phages possess special enzymes capable of degrading these biofilms, exposing and eradicating the bacteria. A 2024 review noted significant successes in using phages for MRSA (methicillin-resistant Staphylococcus aureus) bone and joint infections.
- Achromobacter and Persistent Lung Infections: At the 2025 Conference on Bacteriophages, researchers highlighted the success of administering phages via intranasal and intravenous delivery routes to treat drug-resistant Achromobacter xylosoxidans in pediatric patients, restoring healthy airway microbiomes and stabilizing lung function.
The Evolutionary Chess Match: Phage-Antibiotic Synergy (PAS)
One of the main criticisms historically leveled against phage therapy is the concept of bacterial resistance. Just as bacteria can evolve to resist antibiotics, they can also evolve to resist phages by altering their surface receptors or deploying defense mechanisms like the famous CRISPR-Cas system (which originally evolved in bacteria precisely to chop up invading viral DNA).
However, modern microbiology has uncovered a spectacular biological loophole: evolutionary trade-offs.
Bacteria have a limited biological budget. To defend against a phage, a bacterium might mutate the specific receptor on its surface that the phage uses to attach. But these surface receptors often serve a crucial function for the bacterium—many of them are the exact same efflux pumps the bacterium uses to spit out antibiotics.
If the bacterium mutates its efflux pump to become invisible to the phage, it loses the ability to pump out the antibiotic. If it retains the efflux pump to survive the antibiotic, it remains a sitting duck for the phage. By utilizing Phage-Antibiotic Synergy (PAS), doctors can trap superbugs in a fatal evolutionary catch-22. Even if the phage doesn't outright kill all the bacteria, it forces the surviving bacteria to drop their antibiotic resistance, rendering old, previously useless antibiotics effective once again.
Next-Gen Weaponry: Synthetic Biology and CRISPR
As we look toward the future, the integration of advanced biotechnology is pushing phage therapy far beyond its natural origins. Natural phages are highly effective, but relying on them requires finding the exact "key" to fit the bacterial "lock" in nature—a process that historically involved scientists painstakingly screening sewage, rivers, and soil samples.
Today, advanced genomic engineering and Artificial Intelligence (AI) are fundamentally changing the landscape.
- CRISPR-Engineered Phages: While bacteria use CRISPR as an immune system, scientists are now arming phages with CRISPR-Cas payloads. In the recent SNIPR001 trial (targeting E. coli strains in urinary tract infections), phages were engineered to not only lyse the bacteria but also to deliver CRISPR/Cas9 machinery specifically programmed to shred the bacteria's essential survival genes. This two-pronged attack ensures rapid, complete eradication.
- Endolysins: Sometimes, introducing a live, replicating virus into the bloodstream isn't ideal. Instead, biotech companies are extracting the active lethal ingredient of the phage—an enzyme called endolysin. Endolysins cut open the bacterial cell wall directly. Because they act so rapidly on fundamental structural components of the bacteria, it is exceptionally difficult for bacteria to develop resistance to them.
- PhageBanks and AI Matching: Organizations are developing massive, globally networked "PhageBanks". When a patient arrives at a hospital with an untreatable infection, a sample of the bacteria is sequenced. An AI algorithm can then rapidly scan the PhageBank to identify the optimal combination of phages to formulate a personalized "phage cocktail". This cocktail targets the bacteria from multiple angles, preventing the superbug from developing resistance to any single phage.
Navigating the Bottlenecks: Regulation, Immunity, and Manufacturing
If phage therapy is so miraculous, why isn't it available at your local pharmacy today? The answer lies in the rigid infrastructure of modern pharmaceutical regulation and the complex biology of the human body.
Regulatory Hurdles: The FDA and the European Medicines Agency (EMA) were built to regulate static, non-living chemical drugs (like aspirin or penicillin). A drug is expected to be identical in every batch, tested extensively in multi-year, multi-million-dollar Phase I, II, and III clinical trials. Phages, however, are biological entities. A single phage strain might only treat a narrow sliver of patients, and because bacteria evolve, the phage formulation might need to be constantly tweaked or combined into adaptive cocktails. Currently, there are no FDA-approved bacteriophage products for general clinical use. However, the landscape is shifting. With approximately 90 clinical trials ongoing globally (41 in the U.S. alone), regulatory bodies are beginning to adopt flexible "adaptive regulatory pathways". Companies like Adaptive Phage Therapeutics are pushing their personalized PhageBank therapies through late-stage trials, paving the way for regulatory frameworks that approve a process of phage matching rather than a static, single drug. The Immune System Dilemma: The human immune system is designed to eliminate foreign viruses. When phages are introduced into the bloodstream, the liver, spleen, and the body's reticuloendothelial system often identify them as invaders and filter them out. If a patient is given multiple doses over weeks, they may develop neutralizing antibodies that destroy the phages before they ever reach the bacteria. To circumvent this, scientists are experimenting with delivery methods—such as intranasal sprays for lung infections, which show rapid bacterial clearance without systemic immune clearance—as well as encapsulating phages in liposomes (microscopic fat bubbles) that act as stealth cloaks to bypass the immune system. Manufacturing Complexity: Unlike brewing vats of chemical antibiotics, creating medical-grade phages requires growing massive amounts of the dangerous pathogenic superbug first, infecting it with the phage, and then purifying the resulting mixture. If the purification isn't perfect, residual bacterial toxins (endotoxins) left in the mixture can cause a fatal immune reaction (septic shock) in the patient. Advancements in Current Good Manufacturing Practice (cGMP) facilities are slowly solving these scalable purification challenges.Beyond Human Medicine: Agriculture, Aquaculture, and the Environment
The battle against antibiotic resistance will not be won in hospitals alone. Nearly 70% of the world's antibiotics are used not on humans, but in agriculture—administered to livestock to promote growth and prevent disease in crowded conditions. This practice transforms farms into massive incubators for superbugs, which eventually leap to humans via contaminated meat, agricultural runoff, and farm workers.
Phages offer a sustainable, non-toxic alternative.
- Agriculture and Food Safety: Phage preparations have already gained regulatory approval in the food industry. Products are routinely sprayed onto deli meats, poultry, and cheeses to eliminate Listeria, Salmonella, and E. coli before they hit grocery store shelves. Because phages do not affect plant or human cells, they leave no toxic residue and do not alter the taste, color, or smell of the food.
- Aquaculture: Fish farming is a vital global food source, but fish raised in dense, aquatic environments are highly susceptible to bacterial outbreaks. Dumping antibiotics into the ocean destroys local ecosystems. Phages can be added to fish feed or water to selectively eliminate pathogens without causing environmental collateral damage.
- Veterinary Medicine: From treating mastitis in dairy cows to combating respiratory infections in poultry, veterinary medicine is rapidly adopting phage technology. Not only does this save animal lives, but it also directly cuts off a major pipeline of antibiotic resistance from trickling into the human domain.
The Road Ahead: Preparing for 2050
As we look toward the ominous 2050 deadline set by the WHO and the United Nations—a year where unchecked antimicrobial resistance could result in up to 39 million more dead and inflict upwards of $3.4 trillion in annual global economic damages—it is clear that the golden age of the "magic bullet" antibiotic is over.
But the end of one era marks the beginning of another. We are transitioning from the blunt-force trauma of broad-spectrum chemicals to the precision of personalized, biological warfare.
The medicine of tomorrow will look vastly different. When you contract a severe infection, a rapid diagnostic test will identify the exact genetic profile of the invading pathogen within minutes. An AI system will formulate a bespoke therapeutic cocktail: perhaps a low dose of a traditional antibiotic partnered with a genetically engineered, CRISPR-enhanced bacteriophage, tailored specifically to corner the bacteria into evolutionary submission.
Bacteriophages are not a magic cure-all, and they are unlikely to completely replace antibiotics. Instead, they represent a powerful, adaptable ally. For billions of years, these viral assassins have been keeping the bacterial world in check. By finally learning to harness their relentless, microscopic power, humanity stands a fighting chance in the war against antibiotic resistance. We are no longer fighting nature; we are finally partnering with it.
Reference:
- https://www.smithsonianmag.com/smart-news/deaths-from-antibiotic-resistant-infections-could-reach-39-million-by-2050-study-suggests-180985095/
- https://www.who.int/publications/i/item/9789240116337
- https://www.who.int/news/item/13-10-2025-who-warns-of-widespread-resistance-to-common-antibiotics-worldwide
- https://www.cdc.gov/media/releases/2025/2025-cdc-report-finds-sharp-rise-in-dangerous-drug-resistant-bacteria.html
- https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
- https://pro-lab.co.uk/2025/04/28/microbiology-news-april-2025/
- https://synapse.patsnap.com/article/how-many-fda-approved-bacteriophage-therapy-are-there
- https://carb-x.org/wp-content/uploads/2024/03/Turner-et-al-2024-Phage.pdf
- https://www.oaepublish.com/articles/mrr.2025.31
- https://pmc.ncbi.nlm.nih.gov/articles/PMC11423923/
- https://www.mdpi.com/1424-8247/18/8/1115
- https://pubmed.ncbi.nlm.nih.gov/40872508/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC12908700/
- https://www.mdpi.com/2079-6382/14/6/581
- https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2023.1250848/full